To evaluate the diagnostic value of CSF lactate (LCSF) for the diagnosis of bacterial meningitis (BM) following neurosurgery, and compare it with other CSF markers.
MethodsProspective study of consecutive neurosurgical postoperative patients admitted to the Intensive Care Unit (ICU) at Maciel Hospital. Patients with clinical suspicion of BM were categorised, according to preset criteria, into 3 groups: (1) proven BM; (2) probable BM, and (3) excluded BM. CSF markers were plotted in a receiver operating curve (ROC) to evaluate their diagnostic accuracy.
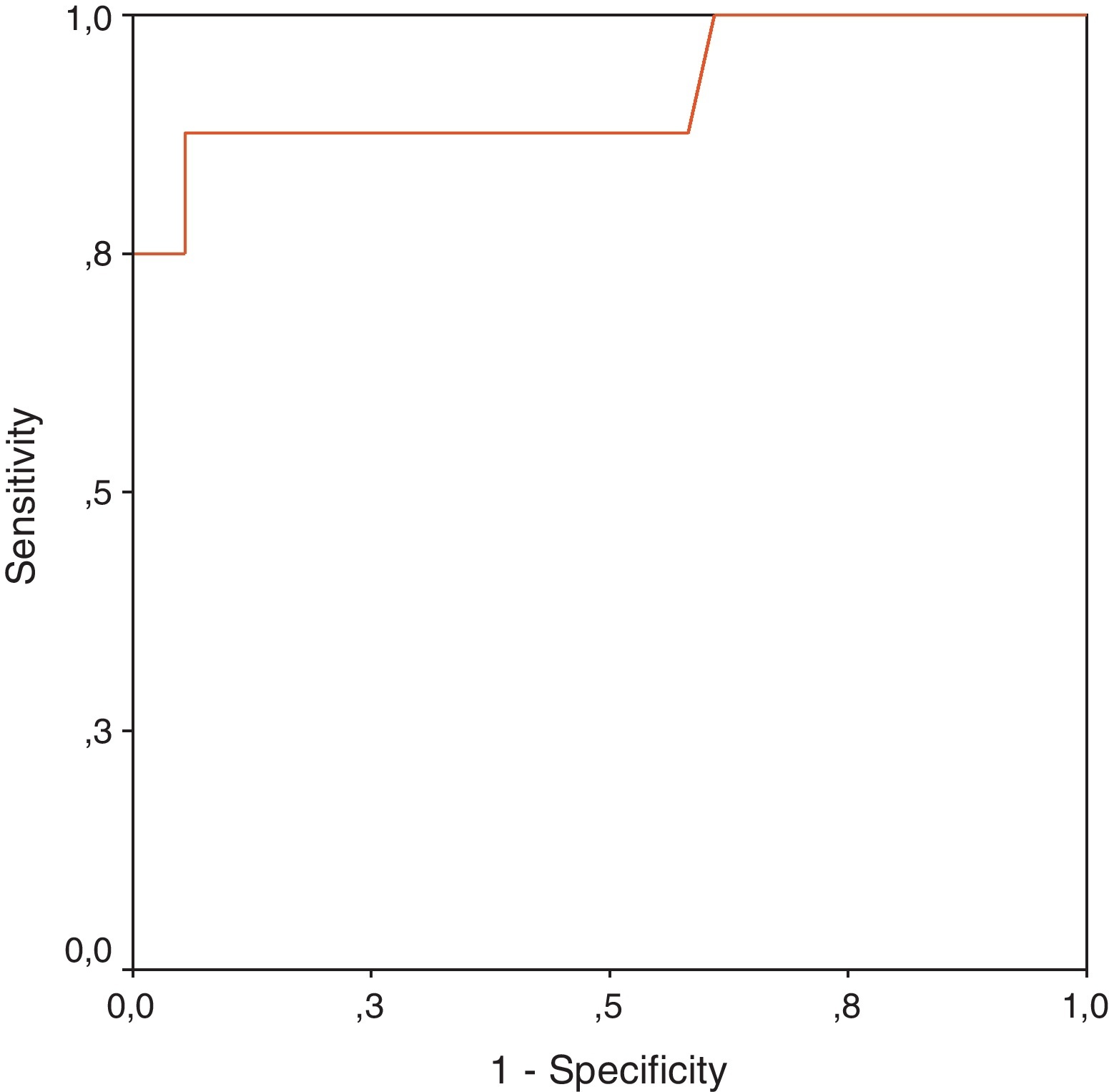
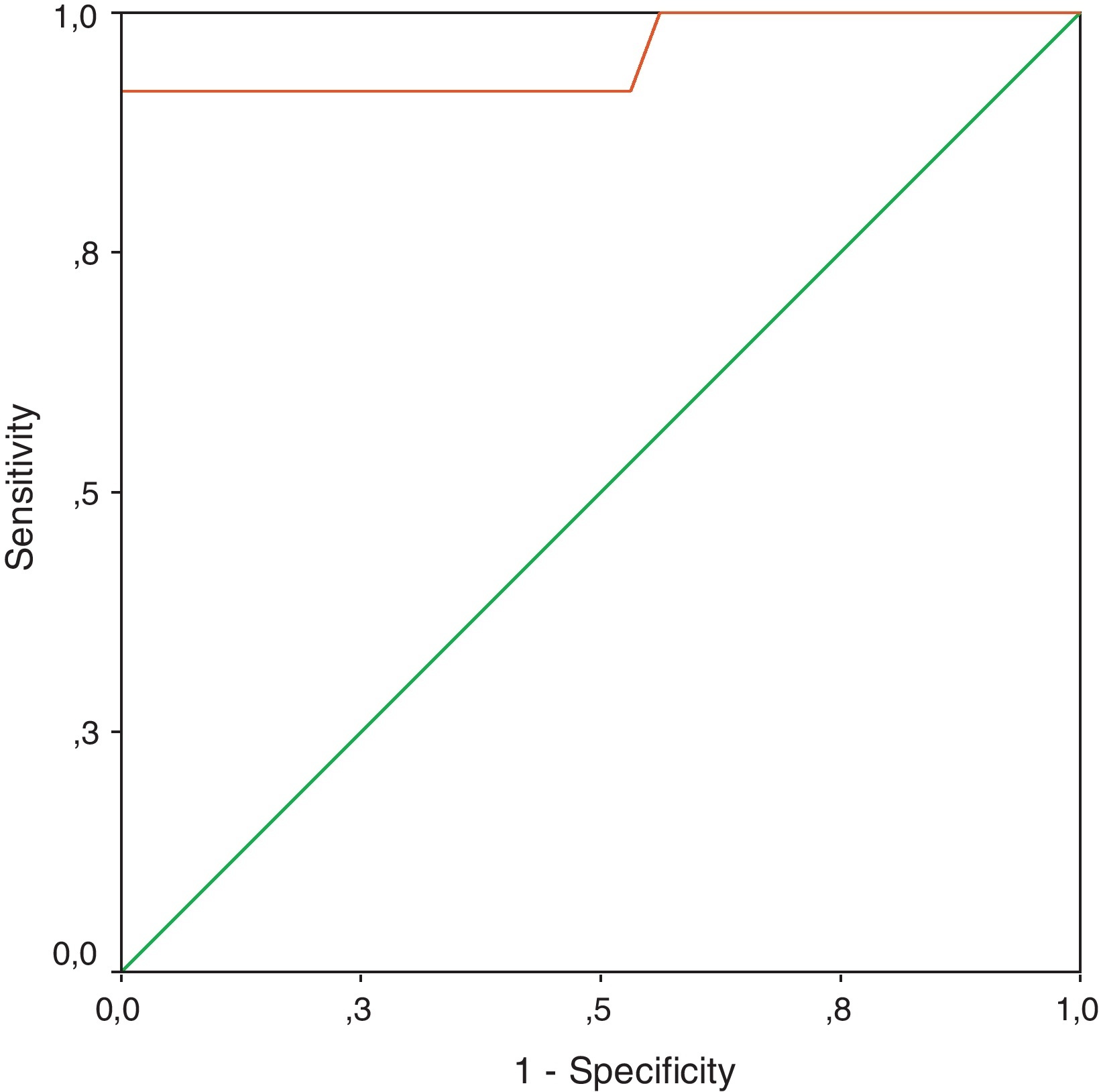
ResultsThe study included 158 patients. We obtained 46 CSF samples from patients with clinical suspicion of BM by lumbar puncture (LP): 10 corresponded to proven BM, 4 to probable BM and 32 to excluded BM. Mean lactate in CSF (LCSF) was: 10.72±4.68mM for proven BM, 6.07±0.66mM for probable BM and 3.06±1.11mM for excluded BM (P<.001 for proven BM and probable BM vs excluded BM; P=NS for proven BM vs probable BM). LCSF displayed a better diagnostic accuracy for BM in the 2 scenarios studied: (1) positive bacterial CSF culture or Gram stain as positive control (gold standard) (sensitivity: 87%, specificity: 94%, cut-off value: 5.9mM), and (2) combination of proven BM and probable BM as positive control (sensitivity: 92%, specificity: 100%, cut-off value: 5.2mM).
ConclusionsAccording to our results, determination of LCSF is a quick, sensitive and specific test to identify the need for antimicrobial therapy in neurosurgical postoperative patients with clinical suspicion of BM.
Evaluar el valor diagnóstico del lactato en líquido cefalorraquídeo (LCR) para el diagnóstico de meningitis bacteriana (MB) después de una neurocirugía, y compararlo con otros marcadores bioquímicos del LCR.
MétodosEstudio prospectivo de pacientes sometidos a neurocirugía admitidos consecutivamente en la Unidad de Cuidados Intensivos (UCI) del Hospital Maciel. Los pacientes con sospecha clínica de MB, fueron categorizados por criterios predeterminados en tres grupos: (1) MB probada, y (2) MB probable, y (3) MB excluida. Los marcadores de LCR fueron analizados de acuerdo a la curva ROC (receiver operating curve) para evaluar su exactitud diagnóstica.
ResultadosSe estudiaron 158 pacientes. 46 presentaron sospecha clínica de MB, de los cuales se obtuvieron muestras de LCR mediante realización de punción lumbar: 10 fueron MB probada, 4 fueron MB probable y 32 MB excluida. La media de lactato en LCR fue: 10,72±4,68mM para MB probada, 6,07±0,66mM para MB probable y 3,06±1,11mM para MB excluida (p<0,0001 para MB probada y MB probable vs MB excluida; p=NS para MB probada vs MB probable). El lactato en LCR demostró la mayor exactitud diagnóstica para MB en los 2 escenarios estudiados: (1) cultivo bacteriano o tinción de Gram positivo en LCR como control positivo (sensibilidad: 87%, especificidad: 94%, valor de corte: 5,9mM); y (2) combinación de MB probada y MB probable como control positivo (sensibilidad: 92%, especificidad: 100%, valor de corte: 5,2mM).
ConclusiónDe acuerdo a nuestros resultados, la medición de lactato en LCR es un método diagnóstico rápido, sensible y específico para identificar la necesidad de iniciar antibioterapia en pacientes con sospecha clinica de MB postquirúrgica.
Article

If it is the first time you have accessed you can obtain your credentials by contacting Elsevier Spain in suscripciones@elsevier.com or by calling our Customer Service at902 88 87 40 if you are calling from Spain or at +34 932 418 800 (from 9 to 18h., GMT + 1) if you are calling outside of Spain.
If you already have your login data, please click here .
If you have forgotten your password you can you can recover it by clicking here and selecting the option ¿I have forgotten my password¿.