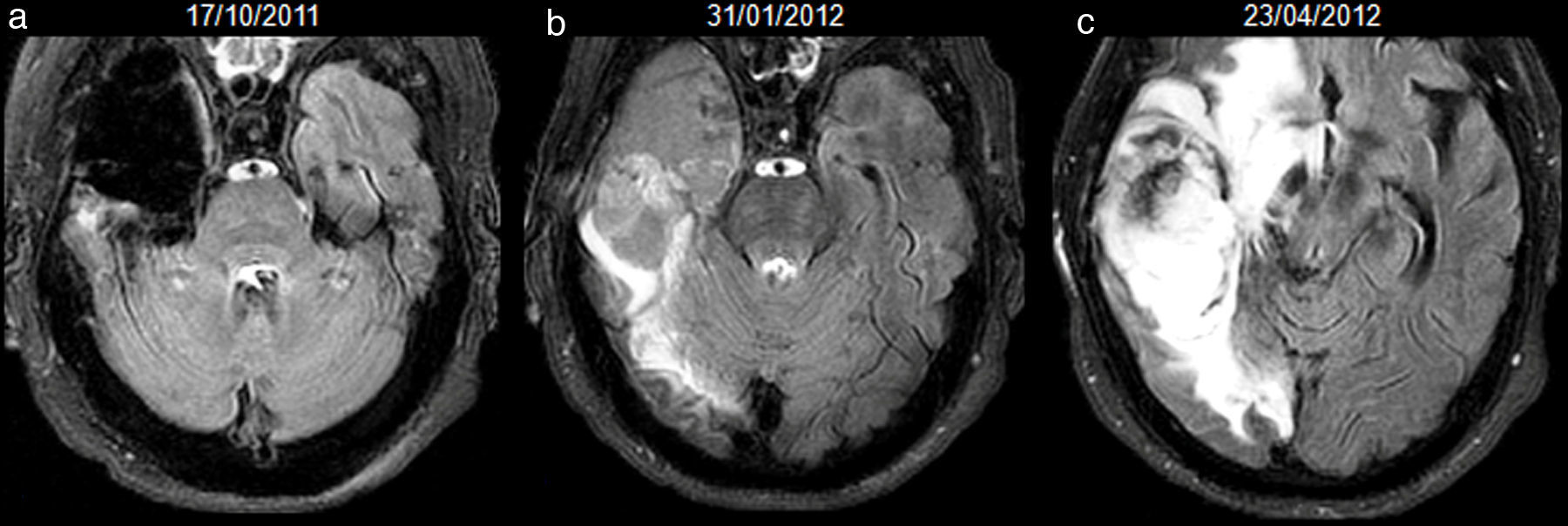
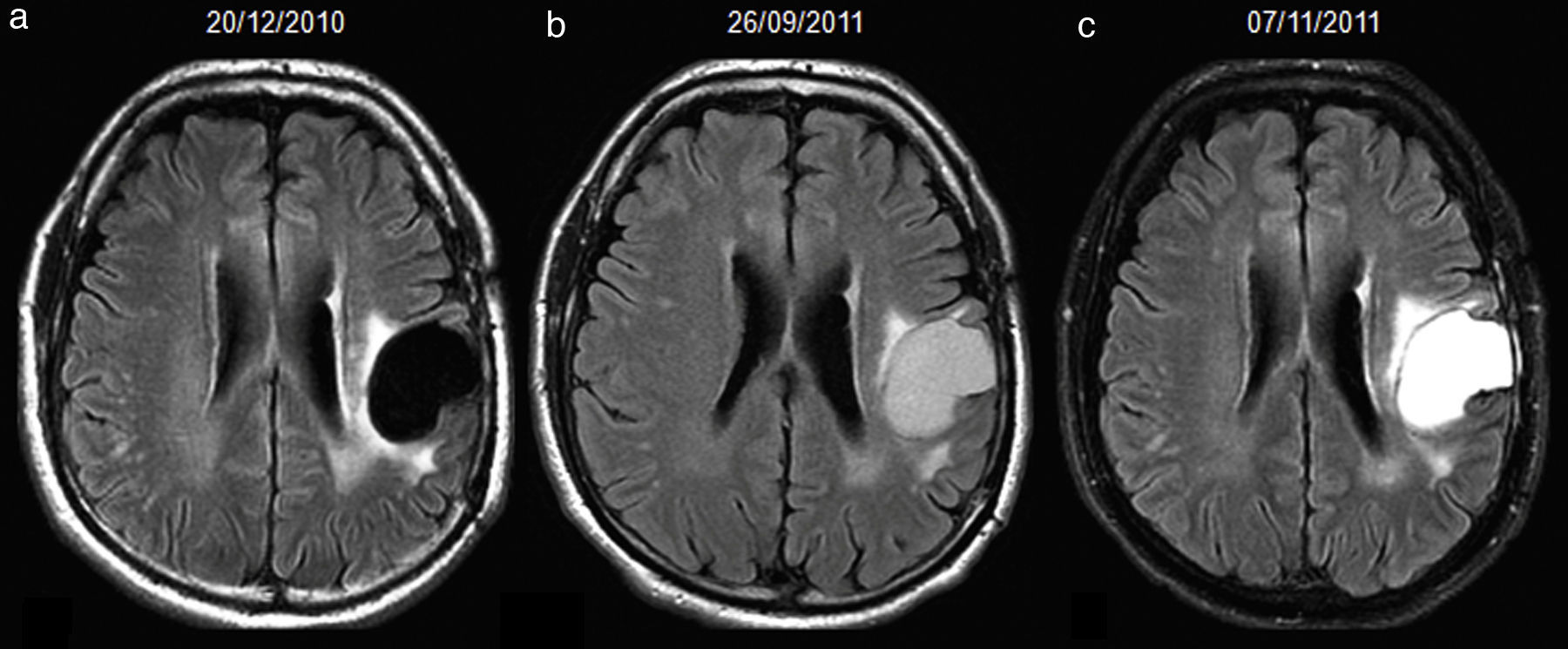
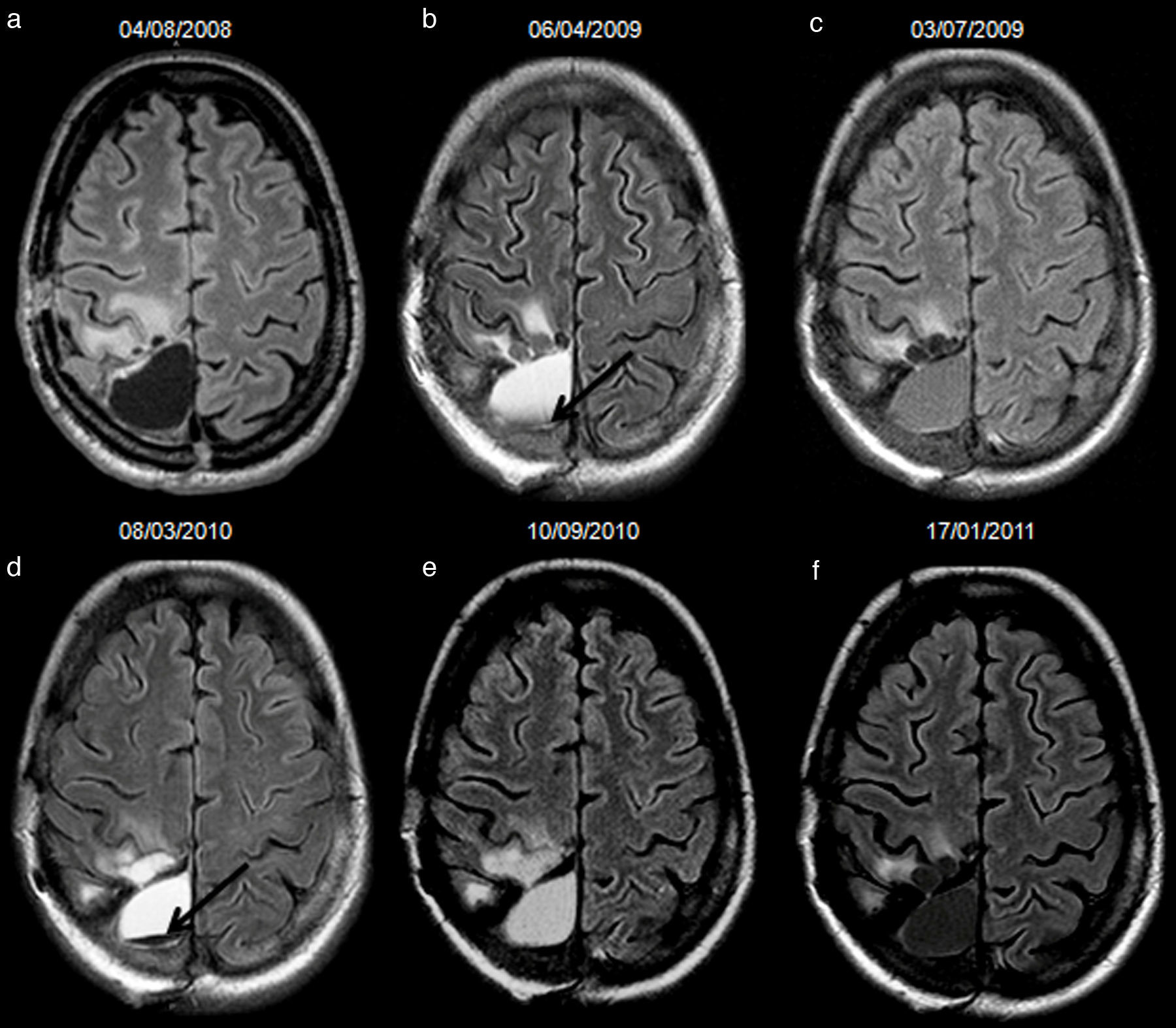
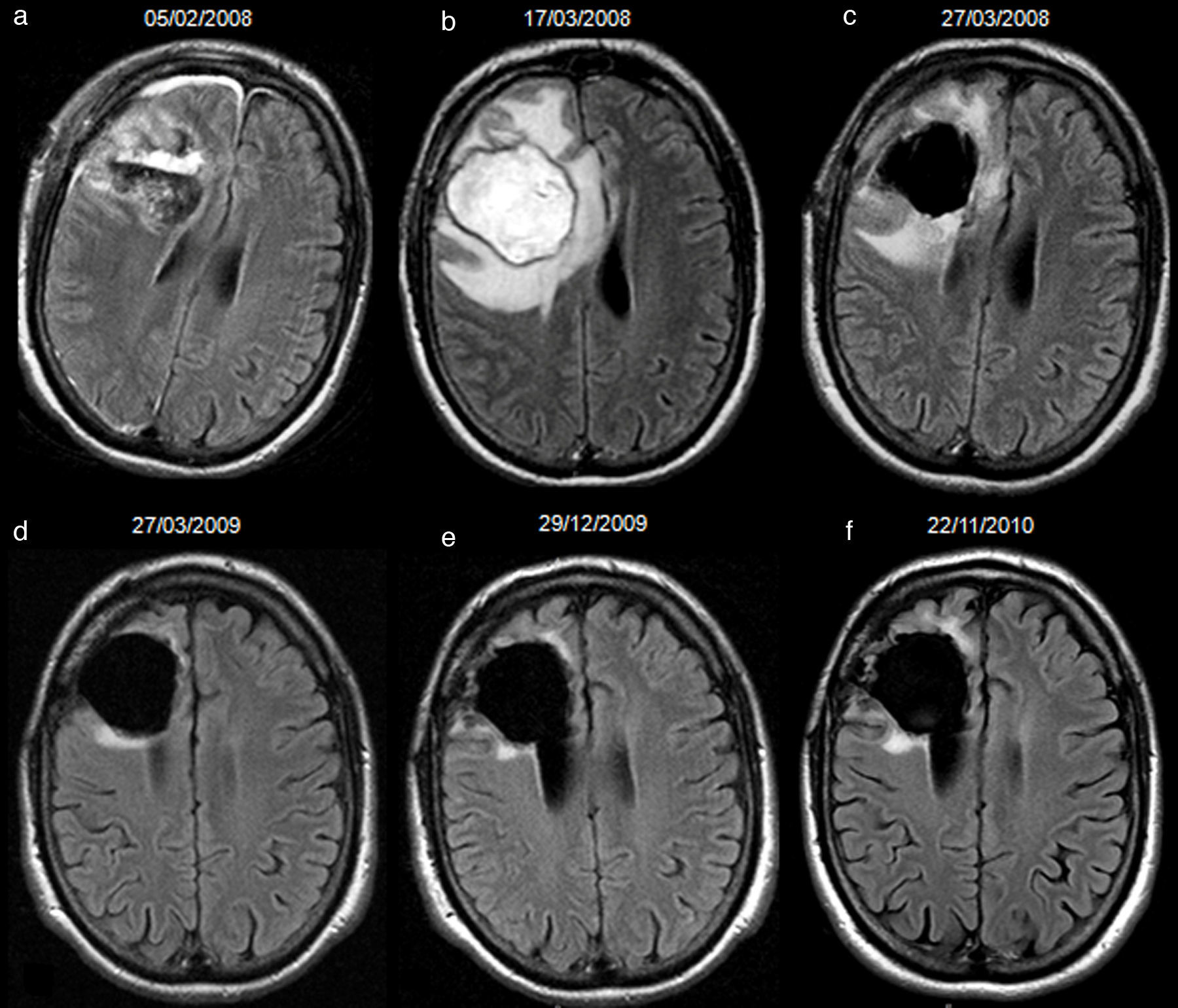
To determine if hyperintense fluid in the postsurgical cavity on follow-up fluid-attenuated inversion recovery (FLAIR) sequences can predict progression in gliomas.
Material and MethodsObservational study of magnetic resonance imaging signal of fluid within the post-surgical cavity in patients with glioma (grade II–IV), with surgery and follow-up between 2007 and 2012. Qualitative comparison between the signal of fluid in the cavity and of the ventricular cerebrospinal fluid (CSF) was performed on FLAIR sequences. Fluid in the cavity was classified as isointense or hyperintense compared to CSF. Double-blind reading was performed. The signal intensity was correlated with tumour progression, assessed using Response Assessment in Neuro-Oncology criteria.
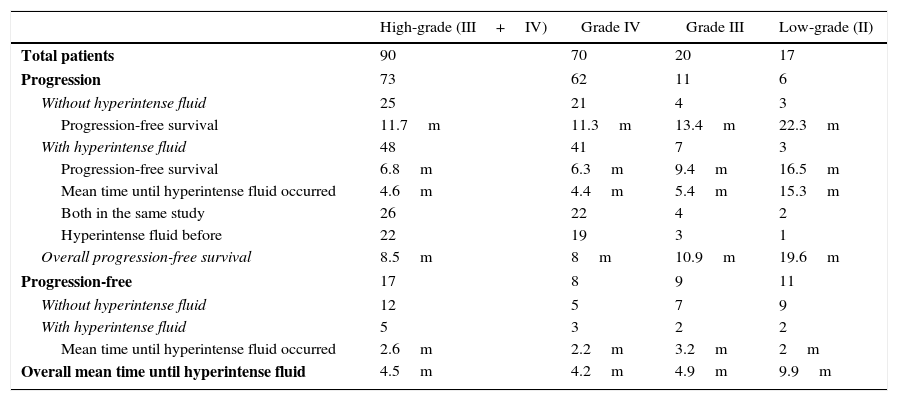
ResultsA total of 107 patients were included, of whom 90 had high-grade gliomas. Inter-rater agreement was excellent, and intra-rater complete (k=0.94 and 1, p<.001). Hyperintense fluid in the resection cavity occurred more commonly (58.9% versus 29.4%, p=.025) and earlier (mean 4.5 versus 9.9 months, p<.001) in high-grade than in low-grade gliomas. Hyperintense fluid was associated with progression in high-grade gliomas, with a sensitivity of 65.7% (95%CI, 54.3–75.6%) and a specificity of 70.6% (95%CI, 46.6–87%), and in low-grade gliomas with a sensitivity of 50% (95%CI, 18.7–81.2%), and a specificity of 81.8% (95%CI, 51.1–96%). The positive predictive value of this sign was 90.6% (95%CI, 79.3–96.3%) for high-grade gliomas, and was higher for grade IV (93.2%, 95%CI, 87.3–99.1%) and lower for grade III (77.8%, 95%CI, 59.6–96%), and low-grade gliomas (60%, 95%CI, 22.9–88.4%). False-positives were identified in 7 patients, due to bleeding or infection. Hyperintense fluid in high-grade gliomas preceded progression in 22 patients (30.1%), with a mean of 4.1 months (SD 2.1, 95% CI, 3.2–5), and associated with poorer progression-free survival (mean 6.8 versus 11.7 months, p=.004).
ConclusionsHyperintense fluid in the resection cavity on follow-up FLAIR sequences occurs more frequently and earlier in high-grade gliomas, and is associated with poorer progression-free survival. Hyperintense fluid is associated with disease progression, and can predict the progression of resected gliomas. False-positives due to bleeding and infection can be observed, and are easily recognizable.
Analizar si la hiperseñal en la cavidad posquirúrgica en secuencia FLAIR puede predecir la progresión en gliomas.
Material y métodosEstudio observacional de la señal en resonancia magnética de la cavidad posquirúrgica en pacientes con glioma (gradoii-iv), con cirugía y seguimiento entre 2007 y 2012. La comparación cualitativa entre la señal del líquido en la cavidad y del líquido cefalorraquídeo (LCR) normal se realizó en las secuencias FLAIR. El líquido en la cavidad se clasificó como isointenso o hiperintenso en comparación con el LCR. Se utilizó una lectura doble ciego. La intensidad de la señal se correlacionó con la progresión tumoral evaluada según los criterios RANO.
ResultadosSe incluyeron 107 pacientes, 90 con gliomas de alto grado. La correlación entre los lectores fue excelente, y la intralector, completa (k=0,94 y 1, p<0,001). La hiperseñal en la cavidad de resección ocurrió con mayor frecuencia (58,9% versus 29,4%, p=0,025) y más temprano (media 4,5 frente a 9,9 meses, p<0,001) en los gliomas de alto grado que en los de bajo grado. La hiperseñal se asoció a la progresión en los gliomas de alto grado con una sensibilidad del 65,7% (IC95%, 54,3-75,6%) y una especificidad del 70,6% (IC95%, 46,6-87%), y en los gliomas de bajo grado con una sensibilidad del 50% (IC95%, 18,7-81,2%) y una especificidad del 81,8% (IC95%, 51,1-96%). El valor predictivo positivo de este signo fue del 90,6% (IC95%, 79,3-96,3%) para los gliomas de alto grado, más alto (93,2%, IC95%, 87,3-99,1%) para los de gradoiv y bajo (77,8%, IC95%, 59,6-96%) para los de gradoiii y para gliomas de bajo grado (60%, IC95%, 22,9-88,4%). En 7 pacientes se identificaron falsos positivos debidos a sangrado o infección. La hiperseñal en la cavidad en gliomas de alto grado precedió la progresión en 22 pacientes (30,1%), con una media de 4,1meses (DE 2,1, IC95%, 3,2-5), y se asoció a peor supervivencia libre de progresión (media 6,8 frente a 11,7meses, p=0,004).
ConclusionesLa hiperseñal en la cavidad de resección en secuencias FLAIR ocurre con más frecuencia y más temprano en gliomas de alto grado, y se asocia a peor supervivencia libre de progresión. La hiperseñal en la cavidad se asocia a la progresión de la enfermedad y puede predecir la progresión de los gliomas operados. Pueden darse falsos positivos debidos a hemorragia e infección, y son fácilmente reconocibles.
Article

If it is the first time you have accessed you can obtain your credentials by contacting Elsevier Spain in suscripciones@elsevier.com or by calling our Customer Service at902 88 87 40 if you are calling from Spain or at +34 932 418 800 (from 9 to 18h., GMT + 1) if you are calling outside of Spain.
If you already have your login data, please click here .
If you have forgotten your password you can you can recover it by clicking here and selecting the option ¿I have forgotten my password¿.